Publications
Dietary fat modulation of lipidomic biomarkers for cardiovascular disease and type 2 diabetes
The consortium partners from DIfE and the University of Reading investigated the molecular relationships of diet, lipid biomarkers and the development of metabolic diseases. The researchers used lipidomics, a modern analytical method, to identify those lipids that are statistically associated with cardiovascular diseases and type 2 diabetes. In addition, the scientists found that a diet with an increased proportion of unsaturated fatty acids leads to a reduction in risk-associated lipids and an increase in low-risk lipids.
The results and complete article can be found here: “Deep Lipidomics in Human Plasma: Cardiometabolic Disease Risk and Effect of Dietary Fat Modulation"
Consumption of dairy food and cardiovascular disease risk
Laury Sellem, a doctoral researcher at the consortium partner UoR, and colleagues from the University Paris Sorbonne Nord in France investigated the associations between the consumption of dairy foods and the risk of cardiovascular disease risk in the French prospective cohort study NutriNet-Santé. They found that despite dairy foods being important sources of dietary saturated fat, their consumption was not associated with overall cardiovascular disease or coronary heart disease risks. However, they observed that high consumption of fermented dairy (e.g. cheese and yogurt) was associated with a decreased risk of cerebrovascular disease (e.g. stroke and transient ischaemic attacks). These results suggest a potential differential effect of specific dairy foods on cardiovascular health.
Read the full article at the following link: "Consumption of dairy products and CVD risk: results from the French prospective cohort NutriNet-Santé".
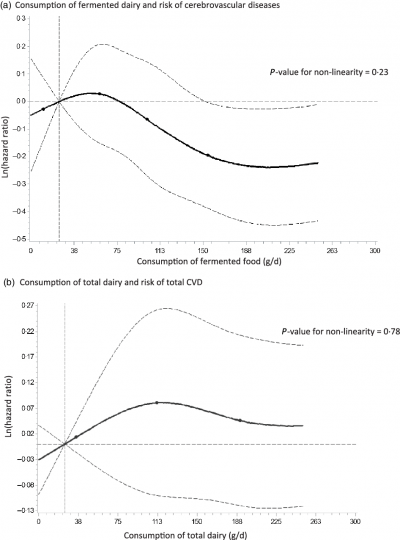
 Estimation;
Estimation;  Upper and lower confidence limit;
Upper and lower confidence limit;  Knots.
Knots.Lipidomics signatures related to heart failure risk
Leveraging the comprehensive lipidomics data in two large human studies, the PREDIMED-trial in Spain and the EPIC-Potsdam cohort in Germany, Wittenbecher, and colleagues from the FAME consortium conducted the first comprehensive prospective lipidomics study on heart failure risk. Single lipids and lipid patterns, particularly ceramides and phosphatidylcholines with palmitate (C16:0), were associated with the risk of developing heart failure in the PREDIMED trial. The prospective associations of several lipids and lipid patterns with heart failure risk were externally validated in the EPIC-Potsdam cohort. These results suggest that plasma lipidomics profiling reflects perturbations in lipid metabolism preceding heart failure onset and generates putative biomarkers of heart failure risk.
Find the full article in the following link "Lipid Profiles and Heart Failure Risk: Results From Two Prospective Studies".
PUFA intake and metabolism and cardiometabolic diseases
FAME investigators discuss in a recent review nutritional implications of the intake and metabolism of omega-3 and omega-6 polyunsaturated fatty acids for cardiometabolic diseases (Schulze et al. Lancet Diabetes Endocrinol 2020). While prospective observational studies support the use of long-chain omega-3 PUFAs in the primary prevention of cardiovascular disease, randomised controlled trials have often reported neutral findings. The harmful effects of a high intake of omega-6 PUFAs are debated, although this idea is not supported by prospective studies. Health effects of PUFAs might be influenced by desaturases, the key enzymes in the metabolism of PUFAs. The activity of these enzymes and modulation by variants of the FADS1-2-3 gene cluster are linked to several cardiometabolic traits.The review discusses the consequences of altered desaturase activity in the context of PUFA intake and their clinical and public health implications.
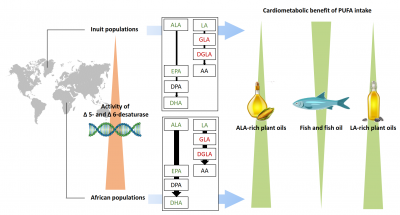
Find the full article in the following link "Intake and metabolism of omega-3 and omega-6 polyunsaturated fatty acids: nutritional implications for cardiometabolic diseases".
List of publications related to the FAME project
- Eichelmann, F., Sellem, L., Wittenbecher, C., Jäger, S., Kuxhaus, O., Prada, M., Cuadrat, R., Jackson, K., Lovegrove, J. and Schulze, M., (2022). Deep Lipidomics in Human Plasma: Cardiometabolic Disease Risk and Effect of Dietary Fat Modulation. Circulation, 146(1), 21-35. https://doi.org/10.1161/CIRCULATIONAHA.121.056805 PubMed
- Prada, M., Wittenbecher, C., Eichelmann, F., Wernitz, A., Kuxhaus, O., Kröger, J., Weikert, C. and Schulze, M., (2022). Plasma Industrial and Ruminant Trans Fatty Acids and Incident Type 2 Diabetes in the EPIC-Potsdam Cohort. Diabetes Care, 45(4), pp.845-853. doi: 10.2337/dc21-1897 PubMed
- Saleh, R.N.M., Minihane, A.M. (2022). Fish, n-3 fatty acids, cognition and dementia risk: not just a fishy tale. Proc Nutr Soc, 81(1):27-40. doi: 10.1017/S0029665121003700 PubMed
- Wittenbecher, C., Cuadrat, R., Johnston, L., Eichelmann, F., Jäger, S., Kuxhaus, O., Prada, M., Del Greco M., F., Hicks, A., Hoffman, P., Krumsiek, J., Hu, F. and Schulze, M. (2022). Dihydroceramide- and ceramide-profiling provides insights into human cardiometabolic disease etiology. Nature Communications, 13(1). doi: 10.1038/s41467-022-28496-1 PubMed
- Wittenbecher C, Eichelmann F, Toledo E, Guasch-Ferré M, Ruiz-Canela M, Li J, Arós F, Lee CH, Liang L, Salas-Salvadó J, Clish CB, Schulze MB, Martínez-González MÁ, Hu FB. (2021). Lipid Profiles and Heart Failure Risk: Results From Two Prospective Studies. Circ Res, 128(3):309-320 doi.org/10.1161/CIRCRESAHA.120.317883 PubMed
- Saleh RNM, West AL, Ostermann AI, Schebb NH, Calder PC, Minihane AM. (2021). APOE Genotype Modifies the Plasma Oxylipin Response to Omega-3 Polyunsaturated Fatty Acid Supplementation in Healthy Individuals. Front Nutr. 8:723813. doi: 10.3389/fnut.2021.723813 PubMed
- Drouin-Chartier JP, Hernández-Alonso P, Guasch-Ferré M, Ruiz-Canela M, Li J, Wittenbecher C, Razquin C, Toledo E, Dennis C, Corella D, Estruch R, Fitó M, Eliassen AH, Tobias DK, Ascherio A, Mucci LA, Rexrode KM, Karlson EW, Costenbader KH, Fuchs CS, Liang L, Clish CB, Martínez-González MA, Salas-Salvadó J, Hu FB. (2021). Dairy consumption, plasma metabolites, and risk of type 2 diabetes. Am J Clin Nutr,114(1):163-174.
doi: 10.1093/ajcn/nqab047. PubMed - Schulze, M., Minihane, A., Saleh, R. & Risérus, U. (2020). Intake and metabolism of omega-3 and omega-6 polyunsaturated fatty acids: nutritional implications for cardiometabolic diseases. The Lancet Diabetes & Endocrinology, 8(11), 915-930. doi.org/10.1016/S2213-8587(20)30148-0 PubMed
- Jäger, S., Cuadrat, R., Hoffmann, P., Wittenbecher, C., & Schulze, M. (2020). Desaturase Activity and the Risk of Type 2 Diabetes and Coronary Artery Disease: A Mendelian Randomization Study. Nutrients, 12(8), 2261. doi.org/10.3390/nu12082261 PubMed
- Curtis, P. J., van der Velpen, V., Berends, L., Jennings, A., Feelisch, M., Umpleby, A. M., . . . Cassidy, A. (2019). Blueberries improve biomarkers of cardiometabolic function in participants with metabolic syndrome-results from a 6-month, double-blind, randomized controlled trial. Am J Clin Nutr, 109(6), 1535-1545. doi:10.1093/ajcn/nqy380 PubMed
- Marklund, M., Wu, J. H. Y., Imamura, F., Del Gobbo, L. C., Fretts, A., de Goede, J., . . . Riserus, U. (2019). Biomarkers of Dietary Omega-6 Fatty Acids and Incident Cardiovascular Disease and Mortality. Circulation, 139(21), 2422-2436. doi:10.1161/circulationaha.118.038908 PubMed
- Muralidharan, J., Papandreou, C., Sala-Vila, A., Rosique-Esteban, N., Fitó, M., Estruch, R., . . . Bulló, M. (2019). Fatty Acids Composition of Blood Cell Membranes and Peripheral Inflammation in the PREDIMED Study: A Cross-Sectional Analysis. Nutrients, 11(3). doi:10.3390/nu11030576 PubMed
- Vallee Marcotte, B., Guenard, F., Lemieux, S., Couture, P., Rudkowska, I., Calder, P. C., . . . Vohl, M. C. (2019). Fine mapping of genome-wide association study signals to identify genetic markers of the plasma triglyceride response to an omega-3 fatty acid supplementation. Am J Clin Nutr, 109(1), 176-185. doi:10.1093/ajcn/nqy298 PubMed
- Li, S. X., Imamura, F., Schulze, M. B., Zheng, J., Ye, Z., Agudo, A., . . . Wareham, N. J. (2018). Interplay between genetic predisposition, macronutrient intake and type 2 diabetes incidence: analysis within EPIC-InterAct across eight European countries. Diabetologia, 61(6), 1325-1332. doi:10.1007/s00125-018-4586-2 PubMed
- Molnos, S., Wahl, S., Haid, M., Eekhoff, E. M. W., Pool, R., Floegel, A., . . . t Hart, L. M. (2018). Metabolite ratios as potential biomarkers for type 2 diabetes: a DIRECT study. Diabetologia, 61(1), 117-129. doi:10.1007/s00125-017-4436-7 PubMed
- Pontifex, M., Vauzour, D., & Minihane, A. M. (2018). The effect of APOE genotype on Alzheimer's disease risk is influenced by sex and docosahexaenoic acid status. Neurobiol Aging, 69, 209-220. doi:10.1016/j.neurobiolaging.2018.05.017 PubMed
- Rathnayake, K. M., Weech, M., Jackson, K. G., & Lovegrove, J. A. (2018). Meal Fatty Acids Have Differential Effects on Postprandial Blood Pressure and Biomarkers of Endothelial Function but Not Vascular Reactivity in Postmenopausal Women in the Randomized Controlled Dietary Intervention and VAScular function (DIVAS)-2 Study. J Nutr, 148(3), 348-357. doi:10.1093/jn/nxx042 PubMed
- Razquin, C., Liang, L., Toledo, E., Clish, C. B., Ruiz-Canela, M., Zheng, Y., . . . Martinez-Gonzalez, M. A. (2018). Plasma lipidome patterns associated with cardiovascular risk in the PREDIMED trial: A case-cohort study. Int J Cardiol, 253, 126-132. doi:10.1016/j.ijcard.2017.10.026 PubMed
- Razquin, C., Toledo, E., Clish, C. B., Ruiz-Canela, M., Dennis, C., Corella, D., . . . Martinez-Gonzalez, M. A. (2018). Plasma Lipidomic Profiling and Risk of Type 2 Diabetes in the PREDIMED Trial. Diabetes Care, 41(12), 2617-2624. doi:10.2337/dc18-0840 PubMed
- Vauzour, D., Rodriguez-Ramiro, I., Rushbrook, S., Ipharraguerre, I. R., Bevan, D., Davies, S., . . . Minihane, A. M. (2018). n-3 Fatty acids combined with flavan-3-ols prevent steatosis and liver injury in a murine model of NAFLD. Biochim Biophys Acta Mol Basis Dis, 1864(1), 69-78. doi:10.1016/j.bbadis.2017.10.002 PubMed
- Wang, D. D., Zheng, Y., Toledo, E., Razquin, C., Ruiz-Canela, M., Guasch-Ferré, M., . . . Hu, F. B. (2018). Lipid metabolic networks, Mediterranean diet and cardiovascular disease in the PREDIMED trial. International Journal of Epidemiology, 47(6), 1830-1845. doi:10.1093/ije/dyy198 PubMed
- Weech, M., Altowaijri, H., Mayneris-Perxachs, J., Vafeiadou, K., Madden, J., Todd, S., . . . Yaqoob, P. (2018). Replacement of dietary saturated fat with unsaturated fats increases numbers of circulating endothelial progenitor cells and decreases numbers of microparticles: findings from the randomized, controlled Dietary Intervention and VAScular function (DIVAS) study. Am J Clin Nutr, 107(6), 876-882. doi:10.1093/ajcn/nqy018 PubMed
- Imamura, F., Sharp, S. J., Koulman, A., Schulze, M. B., Kroger, J., Griffin, J. L., . . . Wareham, N. J. (2017). A combination of plasma phospholipid fatty acids and its association with incidence of type 2 diabetes: The EPIC-InterAct case-cohort study. PLoS Med, 14(10), e1002409. doi:10.1371/journal.pmed.1002409 PubMed
- Jackson, K. G., Lockyer, S., Carvalho-Wells, A. L., Williams, C. M., Minihane, A. M., & Lovegrove, J. A. (2017). Apolipoprotein E (epsilon) genotype has a greater impact on apoB-48 than apoB-100 responses to dietary fat manipulation-insights from the SATgenepsilon study. Mol Nutr Food Res, 61(4). doi:10.1002/mnfr.201600688 PubMed
- Jannasch, F., Bedu-Addo, G., Schulze, M. B., Mockenhaupt, F. P., & Danquah, I. (2017). Serum phospholipid fatty acids, dietary patterns and type 2 diabetes among urban Ghanaians. Nutr J, 16(1), 63. doi:10.1186/s12937-017-0286-x PubMed
- O'Neill, C. M., & Minihane, A. M. (2017). The impact of fatty acid desaturase genotype on fatty acid status and cardiovascular health in adults. Proc Nutr Soc, 76(1), 64-75. doi:10.1017/s0029665116000732 PubMed
- Slim, K. E., Vauzour, D., Tejera, N., Voshol, P. J., Cassidy, A., & Minihane, A. M. (2017). The effect of dietary fish oil on weight gain and insulin sensitivity is dependent on APOE genotype in humanized targeted replacement mice. Faseb j, 31(3), 989-997. doi:10.1096/fj.201600921RR PubMed
- Toledo, E., Wang, D. D., Ruiz-Canela, M., Clish, C. B., Razquin, C., Zheng, Y., . . . Martinez-Gonzalez, M. A. (2017). Plasma lipidomic profiles and cardiovascular events in a randomized intervention trial with the Mediterranean diet. Am J Clin Nutr, 106(4), 973-983. doi:10.3945/ajcn.116.151159 PubMed
- Weitkunat, K., Schumann, S., Nickel, D., Hornemann, S., Petzke, K. J., Schulze, M. B., . . . Klaus, S. (2017). Odd-chain fatty acids as a biomarker for dietary fiber intake: a novel pathway for endogenous production from propionate. Am J Clin Nutr, 105(6), 1544-1551. doi:10.3945/ajcn.117.152702 PubMed
- Wu, J. H. Y., Marklund, M., Imamura, F., Tintle, N., Ardisson Korat, A. V., de Goede, J., . . . Mozaffarian, D. (2017). Omega-6 fatty acid biomarkers and incident type 2 diabetes: pooled analysis of individual-level data for 39 740 adults from 20 prospective cohort studies. Lancet Diabetes Endocrinol, 5(12), 965-974. doi:10.1016/s2213-8587(17)30307-8 PubMed
- Zheng, J. S., Sharp, S. J., Imamura, F., Koulman, A., Schulze, M. B., Ye, Z., . . . Wareham, N. J. (2017). Association between plasma phospholipid saturated fatty acids and metabolic markers of lipid, hepatic, inflammation and glycaemic pathways in eight European countries: a cross-sectional analysis in the EPIC-InterAct study. BMC Med, 15(1), 203. doi:10.1186/s12916-017-0968-4 PubMed
- Forouhi, N. G., Imamura, F., Sharp, S. J., Koulman, A., Schulze, M. B., Zheng, J., . . . Wareham, N. J. (2016). Association of Plasma Phospholipid n-3 and n-6 Polyunsaturated Fatty Acids with Type 2 Diabetes: The EPIC-InterAct Case-Cohort Study. PLoS Med, 13(7), e1002094. doi:10.1371/journal.pmed.1002094 PubMed
- McManus, S., Tejera, N., Awwad, K., Vauzour, D., Rigby, N., Fleming, I., . . . Minihane, A. M. (2016). Differential effects of EPA versus DHA on postprandial vascular function and the plasma oxylipin profile in men. J Lipid Res, 57(9), 1720-1727. doi:10.1194/jlr.M067801 PubMed
- Tejera, N., Vauzour, D., Betancor, M. B., Sayanova, O., Usher, S., Cochard, M., . . . Minihane, A. M. (2016). A Transgenic Camelina sativa Seed Oil Effectively Replaces Fish Oil as a Dietary Source of Eicosapentaenoic Acid in Mice. J Nutr, 146(2), 227-235. doi:10.3945/jn.115.223941 PubMed